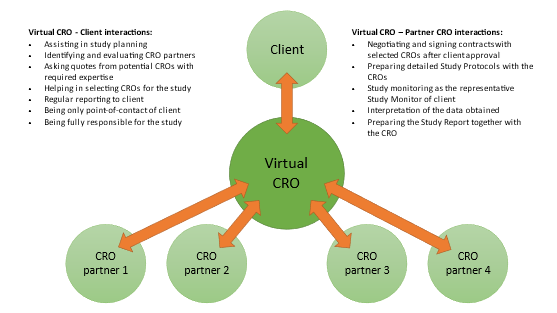
OncoBone co-presented in a webinar titled ‘Working with CROs – from selection to managing relationships’ hosted by Biocom on March 30, 2021. OncoBone’s talk covered CRO selection, and featured speakers from Inotiv and Aligos Therapeutics continued about partnerships between CROs and Sponsors. Below is a summary of the key aspects covered in the webinar.
There are over 3 000 CROs globally and the number in increasing. The market size is estimated to grow at 12% annual rate and reach 45 billion USD by 2022. About 80% of pharma and biotech representatives see that their use of CRO services is likely to increase.
CROs include large CROs that offer a wide variety of models and services, and niche-providers specialized in certain models or services that are not commonly available. With such large variety and choices, how do you know who to select as your CRO partner? And furthermore, how to invest in a relationship that lasts and brings value to the Sponsor? These topics were discussed on March 30, 2021, in a webinar titled: ‘Working with CROs – from selection to managing relationships’.
Featured speakers in the webinar covered aspects from CRO selection to managing relationships between the CRO and the Sponsor. The speakers represented three different industry sectors. Tiina Kähkönen, Chief Scientific Officer at OncoBone represented a consultant offering CRO selection and management as service, Joe Flynn, Chief Commercial Officer at Inotiv represented a CRO, and Dinah Misner, Senior Director at Aligos Therapeutics represented a pharma company working tightly with CROs.
From selection…
Tiina Kähkönen started the webinar by explaining how to identify eligible CROs that can be considered as a partner in preclinical development. CROs can be found from the internet via web searches or from events/conferences, but most commonly new CROs are referred by colleagues. Referrals typically come from colleagues that have been pleased with the good work of a CRO and the overall client experience that they gained, and these are the key factors for committing to work with the same CRO again.
Joe Flynn referred to a survey which showed that 57% of clients stop buying because they had a better experience elsewhere, and actually 90% of clients say that the experience is as important as the product itself. This also speaks to the fact that while there are many good CROs available that can meet the client’s needs in terms of quality and price, other aspects in the overall service become important when committing to work with a CRO again.
Successful working history can transform the relationship between the Sponsor and the CRO, and the CRO may become a preferred partner for the Sponsor. Dinah Misner discussed Preferred Partner Agreements (PPAs) and their relevance to a pharma company. Issues she saw most important in such agreements were related to the CROs capability to accommodate requested timelines, as well as price reductions based on the quantity of studies that the Sponsor purchases from the CRO. In Dinah’s opinion the PPAs also bring value to CROs as pharma clients are more willing to share their internal timelines and plan their research activities for the following years, which can make it easier for the CROs to predict their operations and gives the CROs the possibility to see through the development process.
One of the questions from the audience was related to how common such PPAs are. Joe speculated that they are rare, with about 5% of clients engaging with such agreements. Joe also explained that over time many clients work with multiple CROs. He explained that this is because clients do not have a clear outsourcing strategy, and on the other hand, CROs may not have a client engagement strategy as they only focus on the projects at hand. Qualifying many CROs can increase flexibility, give access to broader expertise, and give the client better scheduling options, but it also adds expenses, dilutes buying power and reduces commitment.
In selecting a CRO, everything starts with the need for a study with certain key parameters that give a response to the research question at hand. For inquiring information, it is often useful for the client to summarize the information for the CRO in a Request For Proposal that contains all essential information that the CRO needs to be able to prepare a quote for the study. Tiina explained that because many CROs can perform the same study, it is advisable to perform a qualification assessment beyond the capability of performing a study. In these qualifications, many Sponsors see value for example in quality, expertise, communication, stability, timelines and price. Sponsors may value certain attributes differently and outlining the importance of these attributes should be done prior to the CRO evaluation.
Audits can be part of the qualification assessment. One of the questions from the audience was related to audits during the pandemic time. Many audits have been arranged virtually. In practice different types of data rooms have been created to securely share data and other documents, and virtual facility tours have been arranged. Joe also pointed out the possibility of using consultants located in the region of the CRO’s premises who could perform the audits on behalf of the Sponsor without the need for the Sponsor to travel to the site.
After the CRO evaluation has been done, you need to visualize and understand the results, and preparing a summary in the form of a scorecard can be useful. In the evaluation you give scores to all CROs that you have evaluated, and if done correctly, the best scoring CRO should be selected for this specific study. It is important that Sponsors adopt a CRO selection process as it helps them to evaluate the CROs for their best interest, and it also helps in starting to build a relationship with the CRO.
…to managing projects and relationships.
Managing projects means monitoring that everything is performed as agreed. In her presentation, Dinah shared the responsibilities of Sponsor and CRO in this process. The Sponsor’s role is to initiate the study and to support the CRO in performing the study. The Sponsor selects a representative to act as a Study Monitor who will be the primary contact for the study, with responsibilities including scheduling, monitoring the in-life phase and reporting. The Study Monitor communicates with the Study Director, the representative responsible for the study at the CRO.
Joe defined the CRO’s role in the client journey as three steps: teach, tailor and take control. In short, this means discussing and aligning with the client on their needs and conducting the study for them. In fact, taking control is probably the best possible word to describe the process as the CRO does take control for one important part of the client’s drug development process. Dinah also highlighted that CROs are key in supporting drug development for many pharma companies, as only a few companies have the resources to conduct all needed studies in-house.
Communication is key to a successful relationship, and when starting the first project together it is important to share as much information as possible, communicate as openly as possible, and not make any assumptions. One of the questions from the audience was related to communications and how to know if the Sponsor’s and CRO’s definitions of certain terms are the same. There is no simple answer to this, but the best way is to ask for clarification and make sure that you are aligned. Wrong interpretation of terminology can lead to mistakes in projects—and mistakes can lead to adversity. Tiina pointed out that mistakes are often inevitable but when they occur, it is important to communicate respectfully and concentrate on problem solving. Joe pointed out that adversity can, in some cases, strengthen the relationship as solving problems together creates trust; but to survive adversity, it is important to commit to the relationship before it happens.
One of the ways on avoiding adversity and building relationships is to work with the same Study Director within the CRO when possible. Dinah also pointed out that the Study Director knows the research program, and can be better prepared to address any challenges and help in interpreting the results beyond normal input from another Study Director. Furthermore, input from CROs and their Study Directors is important as they have a long history of working, for example, with certain research models and classes of compounds, which helps them in providing insight beyond a specific study.
Investing in relationships is important also from the perspective of a CRO. Joe explained that for a CRO it is 5 times more expensive to get a new client than to keep an existing client, and 71% of clients end the relationship due to bad experience. It’s important for a CRO to know what their clients think of their service and if they are satisfied or not. Joe mentioned that one option is to collect Voice Of Client feedback, which is a good way to improve CRO action. It is also important to follow up with clients for increasing satisfaction and to be able to improve the client’s experience when working with the CRO. Investing in a relationship helps both the CRO and Sponsor in project execution, improves communication and lowers expenses.
Concluding remarks
Adopting a CRO selection process helps in finding valuable partners and managing relationships for the best possible outcome. From a client perspective, expertise and experience are key factors in selecting a CRO, and from a CRO perspective, working together to build trust and strong relationships helps to serve clients better and build continuity of their business.
If you were unable to attend and would like to hear the full webinar, a recording can be found from Biocom’s webpages from the following link.